Join us for an event September 26!
How to Streamline RNA-Seq analysis and increase productivity—point, click, and done
Page History
...
The Trim tags task allows you to process unaligned read data with adaptors, barcodes, and UMIs using a Prep kit file that specifies the cofiguration of these elements in your NGS reads.
Running Trim tags
- Click an Unaligned reads data node
- Click the Pre-alignment QA/QC section of the toolbox
- Click Trim tags
...
- Choose a Prep kit from the drop-down menu
Click Finish to run Trim tags (Figure 1)
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
...
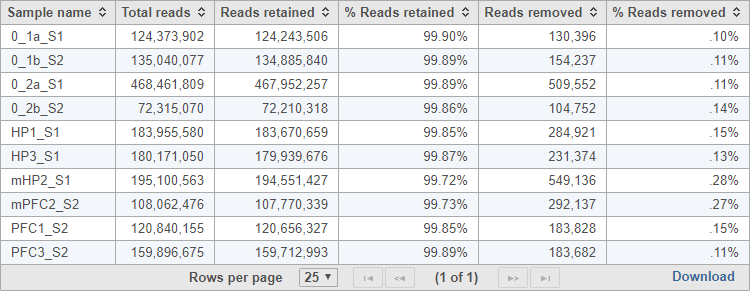
The task report provides a table with the total reads, reads retained, % reads retained, reads removed, and % reads removed for each sample (Figure 2). You can click Download at the bottom of the table to save a text file copy to your computer.
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
Building a custom prep kit
- Select Other / Custom from the Prep kit name drop-down menu
- Give the new prep kit a name
- Choose Build prep kit
...
The Prep kit builder interface will load (Figure 4).
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
...
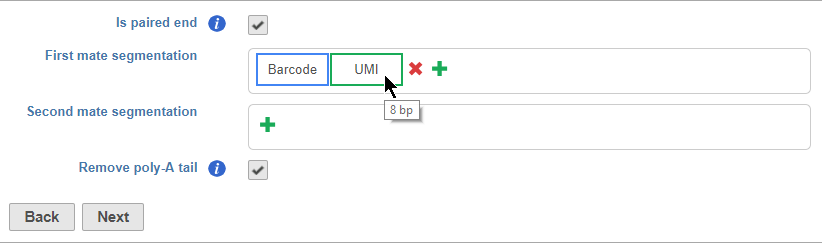
Is paired end - select to switch from single end to paired end FASTQ files (Figure 5). If you choose paired end, the First mate will correspond to the _R1 FASTQ file and the Second mate will correspond to the _R2 FASTQ file.
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
...
Segments include adaptors, barcodes, UMIs, and the insert (i.e., the target sequence of the assay)
Adaptors
For adaptors, you have the option of choosing a file with your adaptor sequences or entering the adaptor sequences manually.
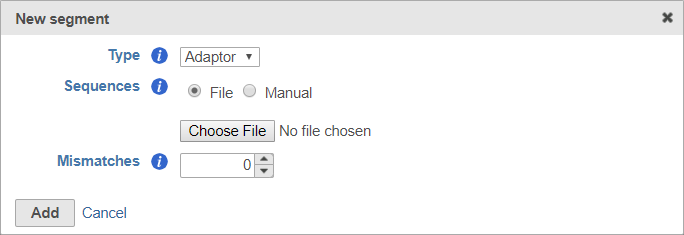
To use a file, choose File for Sequences and then click Choose File (Figure 6). Use the file browser to choose a FASTA file from your local computer.
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
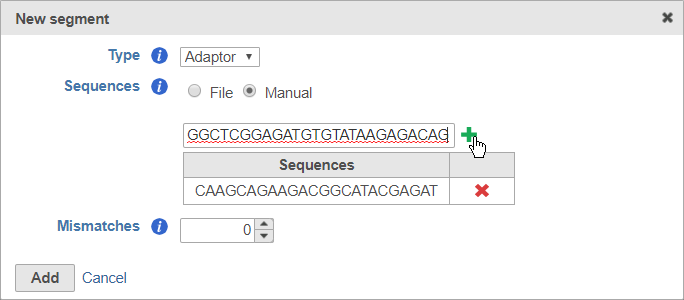
To enter the sequences manually, choose Manual for Sequences then type or paste the adaptor sequences into the text field and click to add the adaptor (Figure 7). You must click for the adaptor sequence to be included. You can remove any adaptor you have added by clicking .
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
...
After you have specified the file or manually entered the sequences, click Add to add the adaptor sequence(s).
UMIs
Unique Molecular Identifiers (UMIs) are randomly generated sequences that uniquely identify an original starting molecule after PCR amplification.
...
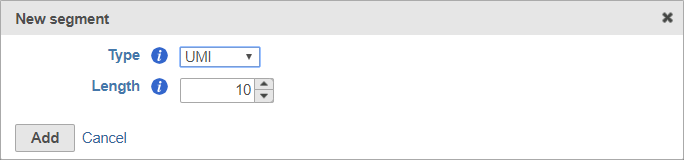
When adding a UMI segment to your prep kit, you can specify the length of your UMIs (Figure 8).
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
Barcode
Adding a barcode segment to a prep kit allows you to access downstream tasks that use barcode information, including Filter barcodes and Quantify barcodes to annotation model (Partek E/M). While the barcode sequence will be trimmed, a record of the barcode sequence for each read is retained for use by downstream tasks.
...
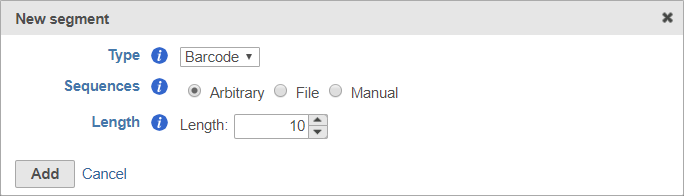
To set the barcode to an arbitrary segment of fixed length, choose Arbitrary and specify the barcode length (Figure 9).
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
Remember to click Add to add the new segment to your prep kit.
Insert
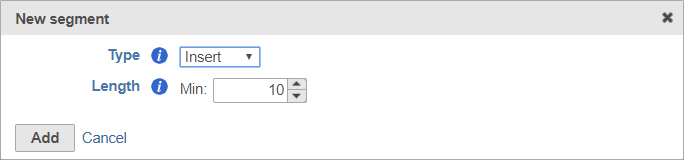
The insert is the sequence retained after trimming in the Trimmed reads data node. For example, in RNA-Seq, this would be the mRNA sequence. Every prep kit must include an insert segment. You can specify the minimum size of the insert section using the Length field (Figure 10). Reads shorter than the minimum length will be discarded.
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
Remember to click Add to add the new segment to your prep kit.
Ordering segments
Segments are placed from 5' to 3' in the read in the order they are added. You should add the 5' segment first and add additional elements in order of their position in the read. Segments will appear in the Segmentation sections as they are added. You can mouse over a segment to view its details (Figure 11).
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
Custom prep kit example
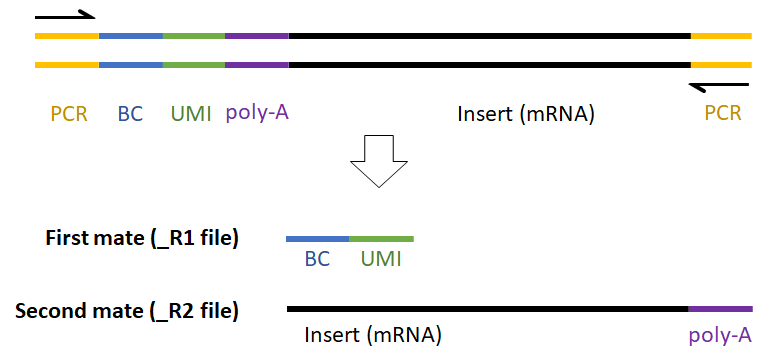
For example, the expected read structure (Figure 12) and a completed prep kit for a standard Drop-seq library prep are shown below (Figure 13).
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
...
- Click Next to complete your prep kit
Managing prep kits
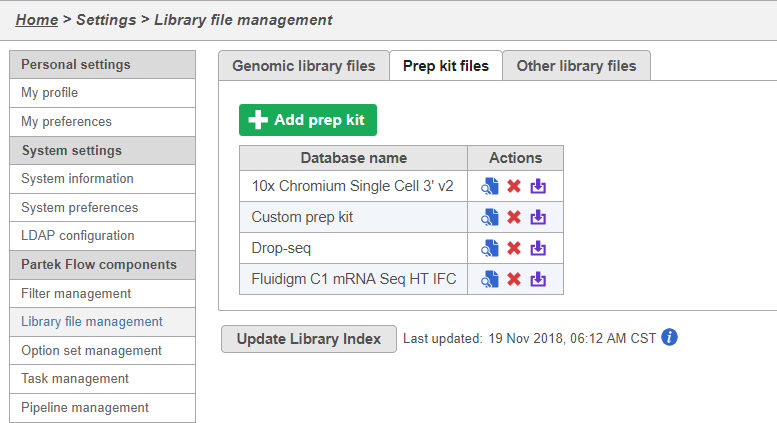
You can manage saved prep kits by going to Home > Settings > Library file management and opening the Prep kit files tab (Figure 14).
| Numbered figure captions | ||||
|---|---|---|---|---|
| ||||
...
Prep kits download as a .zip file. This Prep kit .zip file can be imported into Partek Flow by selecting Import from a file when adding a new prep kit. Select the .zip file when importing, do not unzip the file.
| Additional assistance |
|---|
| Rate Macro | ||
|---|---|---|
|
...